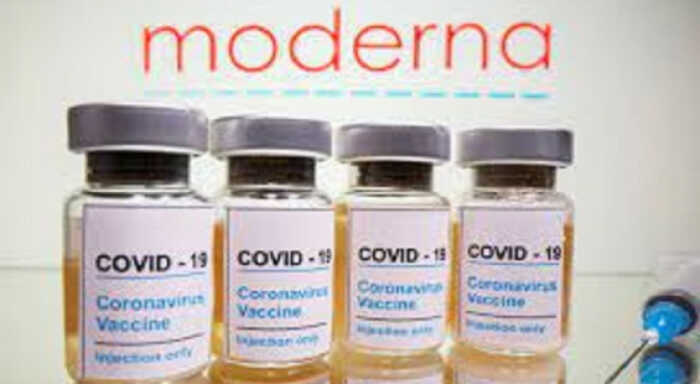
Brussels, March 4 ( /Xinhua) The European Medicines Agency (EMA) has authorised Moderna’s coronavirus vaccine for children aged 6 to 11. EMA’s Head of Biological Health Threats and Vaccines Strategy, Marco Cavaleri said at a press briefing on Thursday that younger children will be given half the dose given to older children and adults. Cavaleri informed that the Moderna shot was also being recommended for use as a booster dose in people who had received other vaccines. Children had previously been given the Pfizer/BioNTech vaccine, which was the only vaccine EMA had approved for children aged five and over. The EMA also recommended booster shots of Pfizer/BioNTech vaccine for those aged 12 and over. Data from countries, including Israel and the ted States, covering over 400,000 children showed that a third dose of the Pfizer/BioNTech vaccine in children aged 12 and over was “safe and effective”, he added. /XINHUA JNA
Bhupendra Singh Chundawat is a seasoned technology journalist with over 22 years of experience in the media industry. He specializes in covering the global technology landscape, with a deep focus on manufacturing trends and the geopolitical impact on tech companies. Currently serving as the Editor at Udaipur Kiran, his insights are shaped by decades of hands-on reporting and editorial leadership in the fast-evolving world of technology.